JAIST researchers innovate new LiB anode material for ultrafast charging
To overcome the slow charging times of traditional lithium-ion batteries (LiB), scientists from the Japan Advanced Institute of Science and Technology (JAIST) have developed a new anode material for lithium-ion batteries (LIBs) that permits ultrafast charging. Produced via a simple, ecologically sound, and efficient approach involving the calcination of a bio-based polymer, this novel material also retained most of its initial capacity over thousands of cycles. A paper on their work is published in the journal Chemical Communications.
One way to shorten the charging time of LIBs is to increase the diffusion rate of lithium ions, which in turn can be done by increasing the interlayer distance in the carbon-based materials used in the battery's anode. While this has been achieved with some success by introducing nitrogen impurities (technically referred to as nitrogen doping), there is no method easily available to control interlayer distance or to concentrate the doping element.
Against this backdrop, a team of scientists from JAIST recently developed an approach for anode fabrication that could lead to extremely fast charging of LIBs. The strategy constitutes a relatively simple, environmentally sound, and highly efficient way to produce a carbon-based anode with a very high nitrogen content.
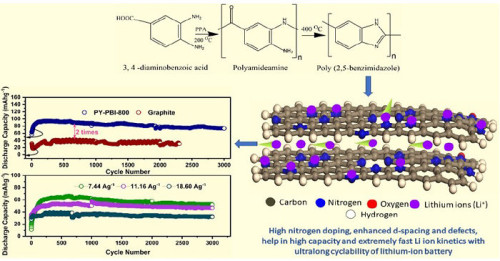
The precursor material for the anode is poly (benzimidazole), a bio-based polymer that can be synthesized from raw materials of biological origin. By calcinating this thermally stable material at 800 °C, the team managed to prepare a carbon anode with a record-setting nitrogen content of 17 percent by weight. They verified the successful synthesis of this material and studied its composition and structural properties using a variety of techniques, including scanning electron tunneling microscopy, Raman spectroscopy, and X-ray photoelectron spectroscopy.
To test the performance of their anode and compare it with the more common graphite, the researchers built half-cells and full-cells and conducted charge-discharge experiments. The results were very promising, as the proposed anode material proved suitable for fast charging, thanks to its enhanced lithium-ion kinetics. Moreover, durability tests showed that the batteries with the proposed anode material retained about 90 percent of initial capacity even after 3,000 charge-discharge cycles at high rates—considerably more than the capacity retained by graphite-based cells.
Another notable advantage of the planned anode material is the use of a bio-based polymer in its synthesis. As a low-carbon technology, the material naturally leads to a synergistic effect that reduces CO2 emissions further.
Modifications to the structure of the polymer precursor could lead to even better performance, which might be relevant for the batteries not only of EVs but also of portable electronics. Finally, the development of highly durable batteries will decrease the global consumption of rare metals, which are non-renewable resources.