Li-ion cell manufacturing: A look at processes and equipment
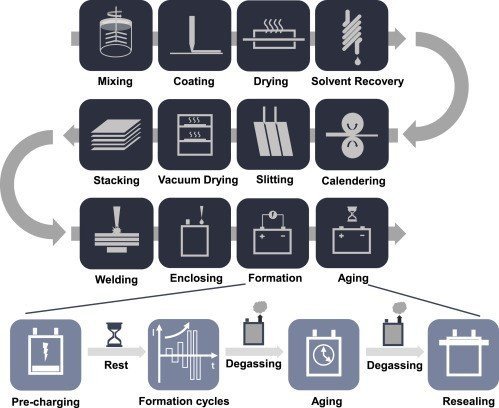
The production of the lithium-ion battery cell consists of three main stages: electrode manufacturing, cell assembly, and cell finishing. Each of these stages has sub-processes, that begin with coating the anode and cathode to assembling the different components and eventually packing and testing the battery cells.
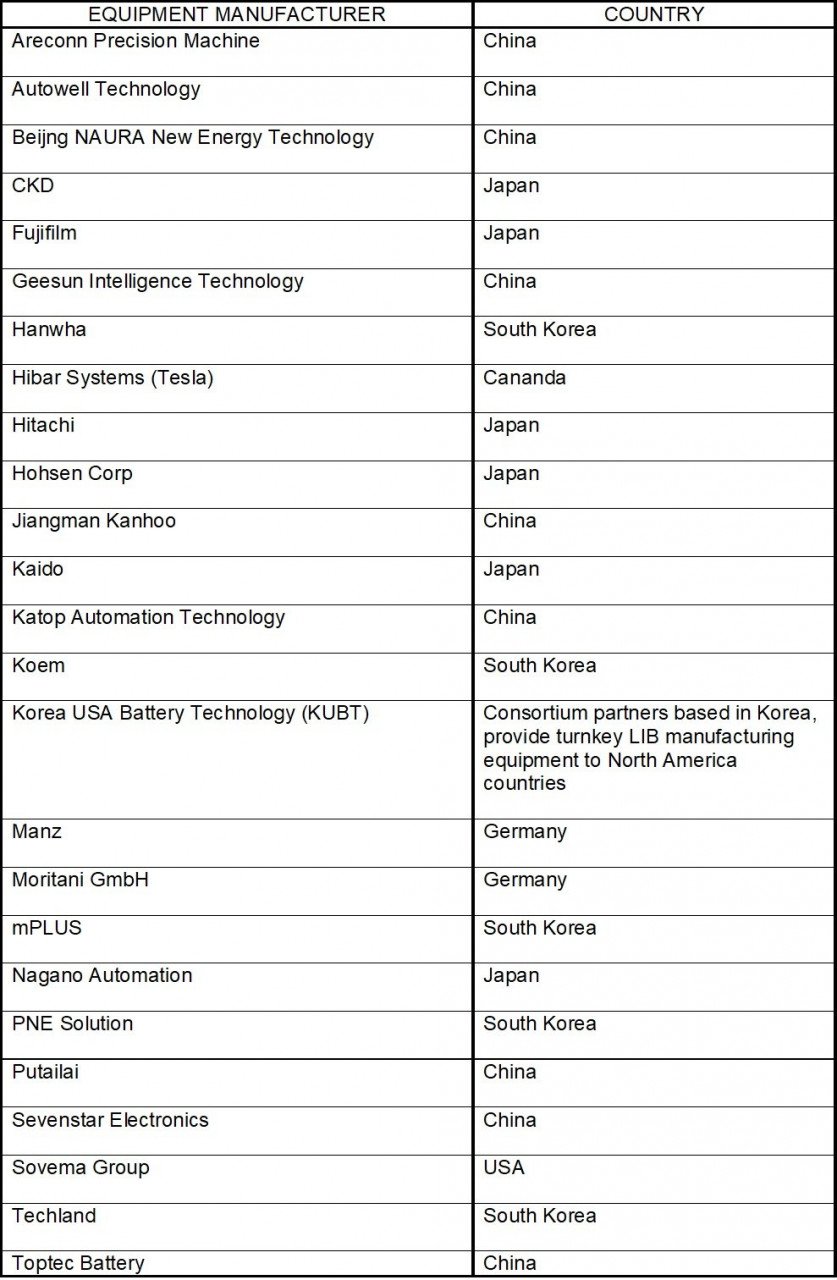
The widely produced types of cells are Prismatic, cylindrical, and pouch, and though the cell design differs the manufacturing processes are more or less similar. Manufacturing contributes about 25 percent of the cost of the Li-ion battery. China, Japan, and South Korea are the major manufacturers and suppliers of equipment for Li-ion cell production.
Li-ion cells comprise four main components – two electrodes: one anode (holds the lithium ions when charged) and one cathode (holds the lithium ions when discharged), a separator that is placed between the electrodes to prevent contact and shorting, and an electrolyte medium that enables movement of lithium ions between the electrodes.
Typically, the anode is made of graphite while the cathode can be an alloy of multiple metals (nickel, cobalt, lithium, others). All the components are packed in a casing and tabs are exposed for a positive and negative terminal. The cells are then arranged and connected to make a battery pack.
STAGE 1 : ELECTRODE MANUFACTURING
Process
The first stage is to mix the electrode materials with a conductive binder to form a uniform slurry with the solvent. (The anode material is a form of Carbon and the cathode is a Lithium metal oxide. To avoid contamination between the two active materials, the anodes and cathodes are usually processed in different rooms.)
The slurry is then coated either continuously or intermittently on both sides of the current collector (Al foil for cathode and Cu foil for the anode) using an application tool (e.g. slot die, doctor blade, anilox roller). The thickness of the electrode coating can be controlled in the coating machine.
The coated foil is fed directly into a long drying oven to evaporate the solvent. (The highly flammable solvent contained in the cathode coating is recovered or used for thermal recycling. From the water-based anode coating, the harmless vapor is exhausted to the ambient environment directly.)
This is followed by the calendaring process where the coated foils are compressed by a rotating pair of rollers. This process helps adjust the physical properties (bonding, conductivity, density, porosity, etc.) of the electrodes.
After calendaring, the finished electrodes are cleaned and fed into slitting machines to be cut into narrow strips and recoiled. The coils are then sent to a vacuum oven to remove the residual moisture and solvent.
(Active materials, conductive additives, solvents, and binders, as well as the aluminum foil and copper foil, are usually purchased components for the cell manufacturer.)
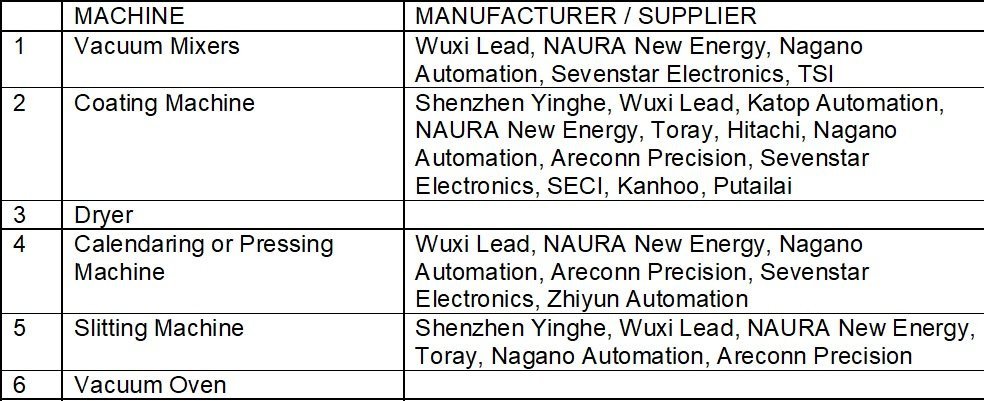
Equipment used in the Process
Machines used in the different processes in the first stage of cell manufacturing include mixers for slurry making, coating and drying machines, calendaring or roll pressing machines, electrode cutting or slitting machines, and vacuum drying ovens.
STAGE 2: CELL ASSEMBLY
Process
After the electrodes have been prepared, they are sent to the dry room for the sub-assembly process where the separator is layered between the anode and the cathode to form the internal structure of the cell. Two basic electrode structures are used depending on the type of cell case: stacking is done for pouch cells, and winding is done for prismatic and cylindrical cells. Cell assembly is usually carried out on highly automated equipment.
In the next step, the assembled cell structure is connected to the terminals or cell tabs, together with any safety devices, using an ultrasonic or laser welding process. The sub-assembly is then inserted into the cell housing (pouch or metal case, depending on the cell design) which is then sealed in a laser welding or heating process leaving an opening for injecting the electrolyte.
This is followed by filling the housed cell with the electrolyte and sealing it. This process is carried out in a dry room since moisture will cause the electrolyte to decompose with the emission of toxic gases. The electrolyte is filled with a high-precision dosing needle. By applying a pressure profile to the cell (supply of inert gas and/or generation of a vacuum in alternating operation), the capillary effect in the cell is activated (wetting the separator).
The finished cell is labeled with a batch or serial number on the case.
(The packaging materials, cell housing, and insulation materials, and electrolyte are usually purchased components for the cell manufacturer.)
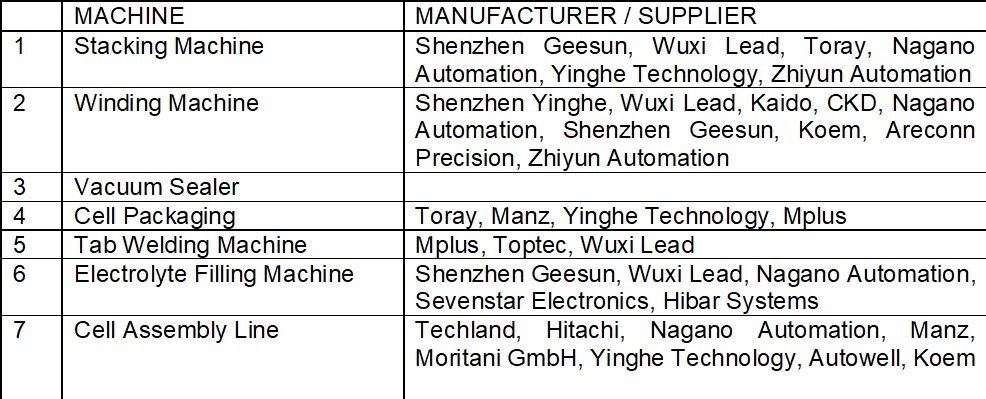
Equipment used in the Process
Machines used in the second stage of cell manufacturing include die cutting machine, stacking machine (pouch cells), winding machine (cylindrical & prismatic), sealing and tab welding machine, and electrolyte filling machine.
STAGE 3: CELL FINISHING
Process
The formation process describes the first charging and discharging processes of the battery cell after the electrolyte is injected into it. The cells are placed in information racks and contacted by spring-loaded contact pins. The cells are then charged or discharged according to precisely defined current and voltage curves. During this process, lithium ions are embedded in the crystal structure of the graphite on the anode side, forming a protective layer called the Solid Electrolyte Interface (SEI) between the electrolyte and the electrode. This protective film results in the low self-discharge of Li-ion batteries and also affects the performance and life of the battery.
(In larger pouch cells, first charging causes strong evolution of gas. This gas can be pressed out of the cell into a dead space called the gas bag. During degassing, the gas bag is pierced into a vacuum chamber and the escaping gases are sucked off. The cell is then finally sealed under vacuum, and the gas bag is separated and disposed of as hazardous waste.)
The next step after formation is aging and is undertaken for quality purposes. During aging, cell characteristics and cell performance are monitored by regularly measuring the open-circuit voltage (OCV) of the cell over a period of up to three weeks. A distinction is made between high temperature (HT) and normal temperature (NT) aging. The cells usually first undergo HT aging and then NT aging. The cells are stored in aging shelves or cabinets. If no significant change is recorded in the cell properties over the entire aging period, it means the cell is fully functional.
After the aging process, the cells are tested in an end-of-line (EOL) test rig. From the aging racks, they are taken to the testing station where they are discharged to the shipping state of charge (capacity measurement). Further pulse tests, internal resistance measurements (DC), optical inspections, OCV tests, and leakage tests can also be carried out.
Once the tests have been completed successfully, the cells can then be assembled in battery packs as per requirement and end-use.
The formation and aging process makes up 32 percent of the total manufacturing process.
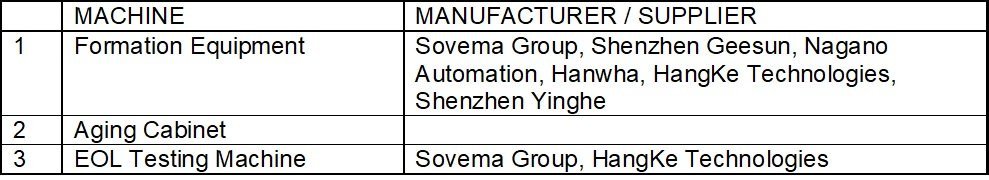
Equipment used in the Process
Machines in the third and final stage of cell manufacturing include battery formation testers/ equipment, aging cabinets, grading machines, and battery testing machines. Generally, coater, winder, and grading & testing equipment account for 70 percent of the total cost of Li-ion cell production equipment, which may vary with the degree of automation.
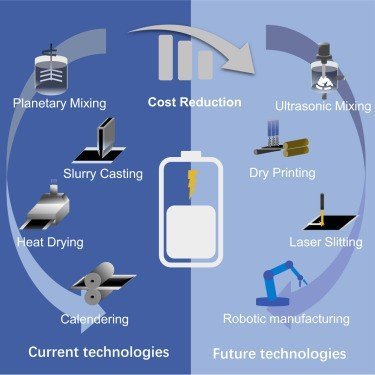
Innovation in Product and Processes
Advancement in technology and materials that are taking place in the field of Li-ion cell manufacturing, will have a positive impact on production costs, reducing the cost of not only raw material but also the final battery cell. Innovation has always led to better performance and quality of the end product.
- Various mixing technologies and mixing tools are now available: intensive mixers, planetary mixers, dispersers, etc.
- Simultaneous coating process that enables coating of the top and bottom sides of the foil at the same time using two opposite application tools.
- Dry coating application of the active material on the carrier foil can be done in powder form without using a solvent, greatly reducing thermal activation time. Removing the solvent and drying process allows large-scale Li-ion battery production to be more economically viable.
- The conventional dryers can be supported by infrared heating, making them more efficient
- Lamination is a key technology for Lithium-ion battery production. The individual electrode and separator sheets are laminated onto each other in a continuous process and are then usually pressed together by a heat press, improving production line speed.